

BDX200
Structure and Composition
BDX200 Proteosomes are a preparation of proteinaceous nanoparticles derived from the Gram-negative bacterium, Neisseria meningitidis. They consist primarily of outer membrane proteins (OMPs), e.g., Classes 1, 2, 4 & Lip, and residual lipo-oligosaccharide (LOS).

Mechanism
TLR 1-2 agonist with some TLR 4 activity. MyD88 dependent.
Meningococcal OMPs engage multiple toll-like receptors. BDX200 is believed to associate non-covalently with hydrophobic domains in amphophilic antigens for presentation to the mucosal immune system. BDX200 should also act directly on the innate immune system independently of an antigen.
Multiple agonists. TLR1-2 & limited TLR4 involvement demonstrated by NF-κB activation in TLR1- 2 and TLR4 transfected HEK cell lines. PV1 activated NF-κB in a concentration dependent manner. BDX200 adjuvant effect on flu immunization (IgG) is reduced in TLR2, TLR4 and MyD88 knock-out mutants of C57BL/6 mice.
Pre-incubation of human peripheral blood mononuclear cells (PBMC) with anti-TLR 1 & 2 antibodies removed the ability of BDX200-influenza to elicit TNF-a and IL-6.
Studies using LPS hypo-responsive (C3H/HeJ) mice suggest that TLR4 does not significantly contribute to the antibody response elicited by BDX200-influenza. However a study of peritoneal macrophages collected from mice and stimulated with BDX200 influenza suggest that TLR4 is involved in the elicitation of TNF-a.
Cytokine Expression
In adherent human mononucleated cells (monocytes vs. non-adherent PBMCs), BDX200 elicited TNF-a, IL-1a, IL-8, RANTES (in some donors), MIP-1a and possibly IL-1b, IL-6, MCP-1 and MIP-1b. Other cell types and tissues targeted by BDX200 are shown in the following table:
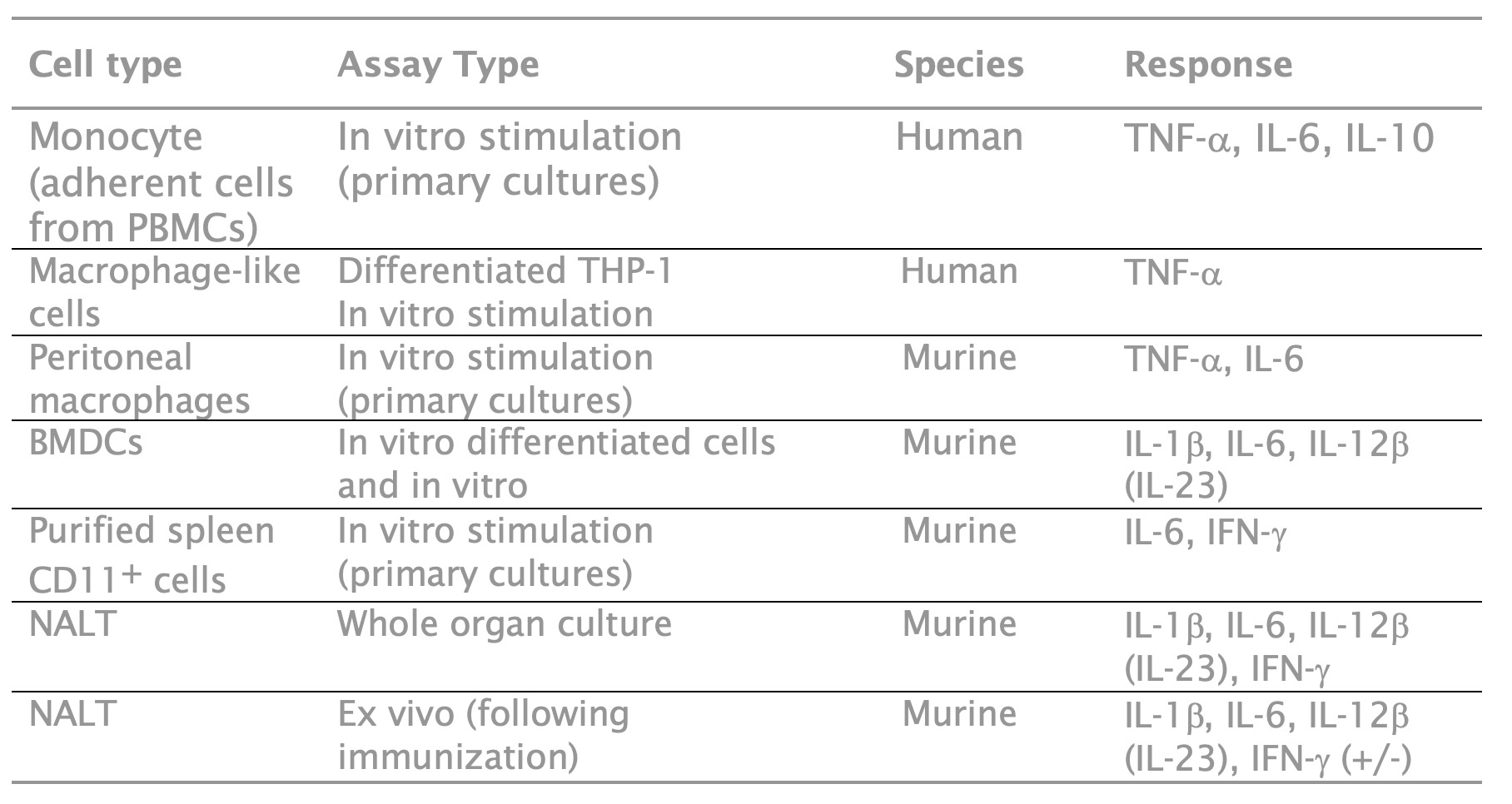
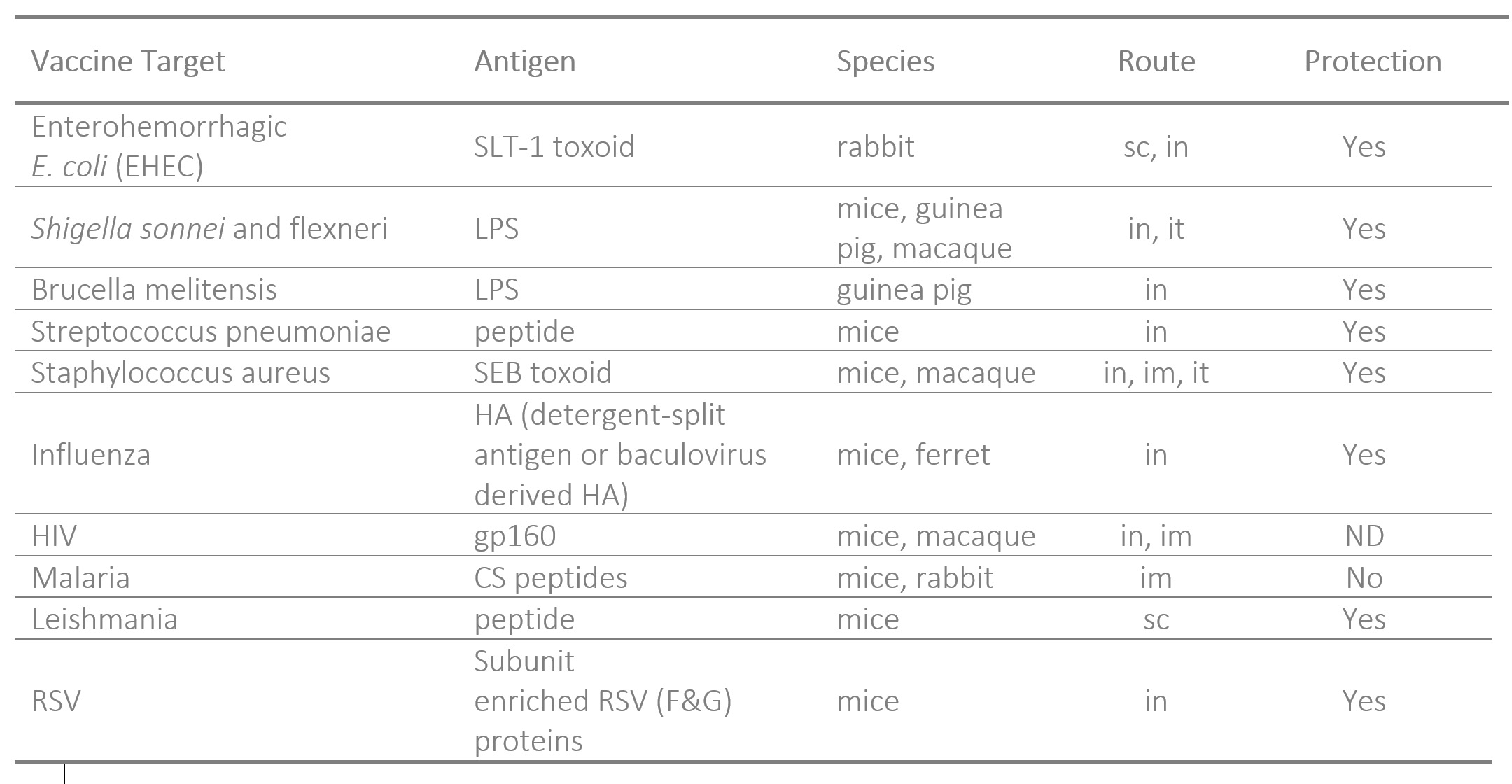
Summary of Preclinical Studies
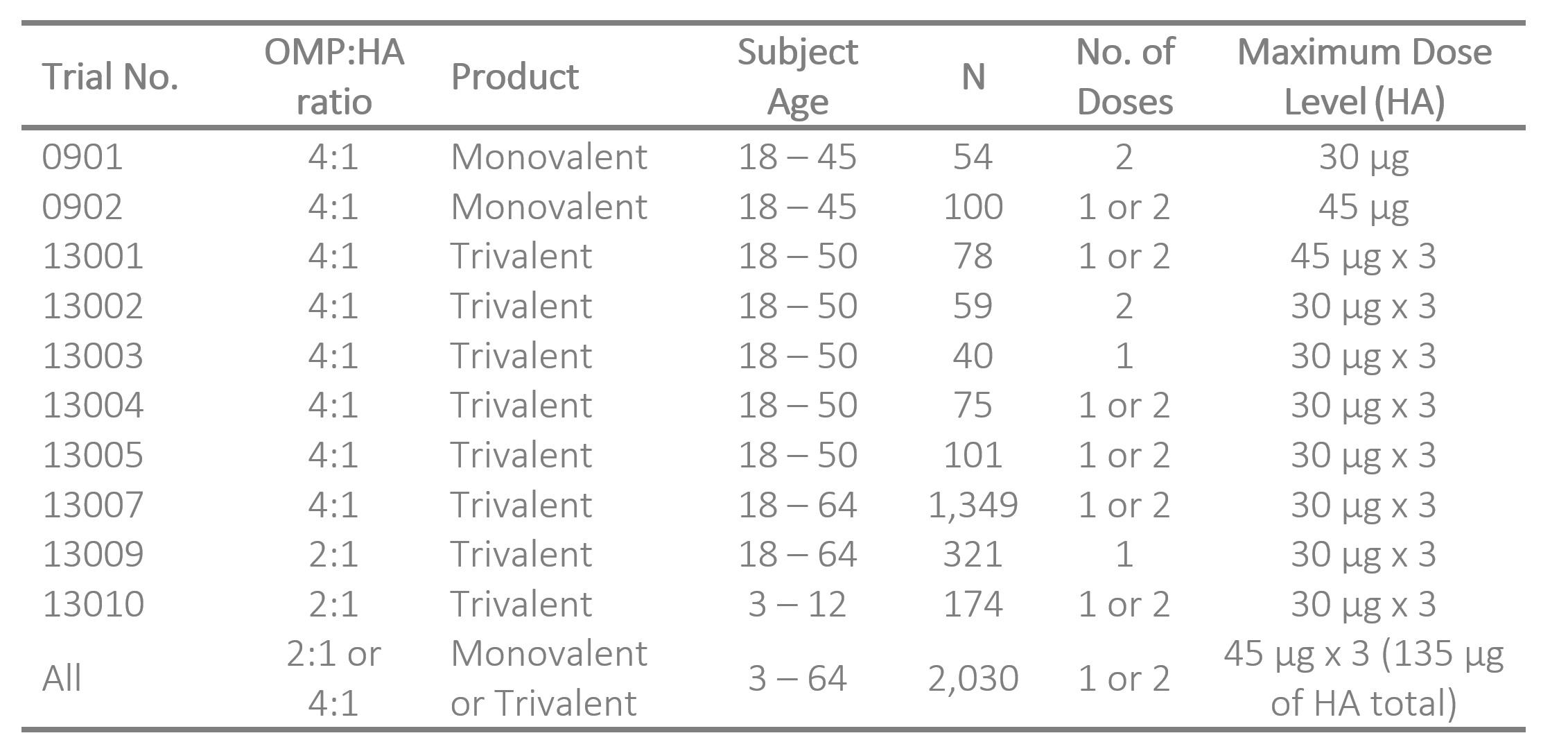
Clinical Trials
Ten clinical trials were conducted on BDX200 complexed to influenza antigen(s). Vaccines were formulated in 2:1 or 4:1 ratios of OMP to HA and administered to healthy volunteers by metered-dose nasal sprays (200 µL total, 100 µL per nostril) either as a single dose, or as two doses at a 14-day interval. Total per dose exposures ranged up to 135 µg HA and ~ 400 to 500 µg OMPs. Trials include one field study for efficacy, two in-patient challenge studies, one reimmunization and one pediatric (3-12 y.o.) study. All trials are summarized below.
Safety
In influenza clinical trials, safety was not statistically different from placebo (trend toward actually fewer respiratory AEs in the vaccine group). In ~2,030 subjects, there were 8 SAEs but only one deemed possibly or probably vaccine-related (mild hypotension with tachycardia eight minutes post-dosing which resolved without sequelae). There were no SAEs in the pediatric trial and reported side effects were comparable to placebo.
Efficacy
BDX200 adjuvanted influenza antigen(s) was compared to placebo in two in-patient challenge studies and in a 2003-04 influenza season field study (all studies had randomized, double-blind, intranasal dosing).
In the challenge trials, two-dose regimens showed 44–61% efficacy against any illness, and 85-100% efficacy against laboratory-confirmed influenza illness. The challenge trials showed relationships between immune response and protection for both serum HAI and salivary IgA, i.e. subjects with highest HAI and IgA levels had best protection against illness, those with lowest levels of each were least protected. Both immunologic parameters were significant predictors of resistance.
The field study in 2003-04 was confounded by an early influenza epidemic, a poor match between the antigen and circulating strains and a low rate of confirmed influenza illness in the study population (~2% placebo rate). Nonetheless, efficacy rates were similar to those of intramuscular, unadjuvanted vaccines in studies carried out in similar drift-variant years.