

Proteosomes
The utility of the Proteosome Nasal Vaccine platform has been established in numerous preclinical animal models of both viral and bac- terial infection. Diseases for which vaccine proof of concept has been demonstrated include; Nasal Vaccines are comprised of components derived from the outer membranes (OMP) of Gram-negative bacteria formulated as nanoparticles, alone or in combination with an antigen. The technology was initially developed at the Walter Reid Army Institute of Technology and later by ID Biomedical Corporation which became part of GSK Vaccines in 2005. Inspirevax has an exclusive license from GSK Vaccines to develop and commercialize this technology.
Proteosome Technology Platform Advantages
Mechanism of Action
OMP formulated with lipo-polysaccharide (LPS) stimulates innate immune responses leading to enhanced adaptive immunity. PorB has been identified as an agonist for TLR-2. LPS and the related lipo-oligosaccharide (LOS) are agonists for TLR- and CD1 [1]. Stimulation of these receptors can increase MHC class I and II expression on antigen-presenting cells, elicit proin- flammatory cytokines that secondarily upregulate the IL-2 receptor, B7.2 (CD86) and adhesion molecules, and stimulate maturation of dendritic cells. Proteosomes also facilitate the uptake of vaccines into mucosal surfaces [2]. Stimulation of both innate and adaptive immunity is ideal for the development of vaccines against pandemic diseases where routine prophylactic immunization is not carried out.
Preclinical Proof of Concept
The utility of the Proteosome Nasal Vaccine platform has been established in numerous preclinical animal models of both viral and bac- terial infection. Diseases for which vaccine proof of concept has been demonstrated include; Influenza, Allergy, RSV, Plague, Anthrax, Group A Strep and Shigella and most recently SARS (Covid-19).
Clinical Experience
Two Nasal Proteosome Vaccines have been studied in clinical trials. A Shigella vaccine comprised of OMP and LPS (S. flexneri) was given to 50 people at doses from 100 to 1,500 mcg. and established the safety of Proteosome Vac- cines in man. A Proteosome Nasal Influenza Vaccine (FluINsure™) was developed by ID Biomedical and studied in 10 clinical trials
Recently, in December 2021, a clinical trial of Protollin as an immunotherapeutic for the treatment of Alzheimer's Disease was initiatied at the Brigham and Women's Hospital (Boston). This is a Phase 1 Ascending Dose Tolerance study in Alzheimer's Disease patients.
Variants
Proteosome adjuvant variants that have been manufactured and studied are described in the following table.
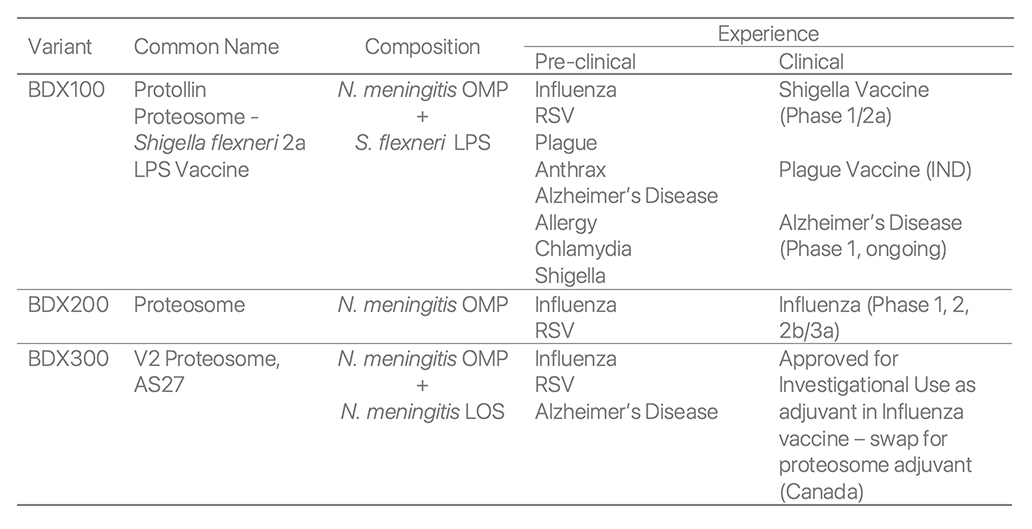
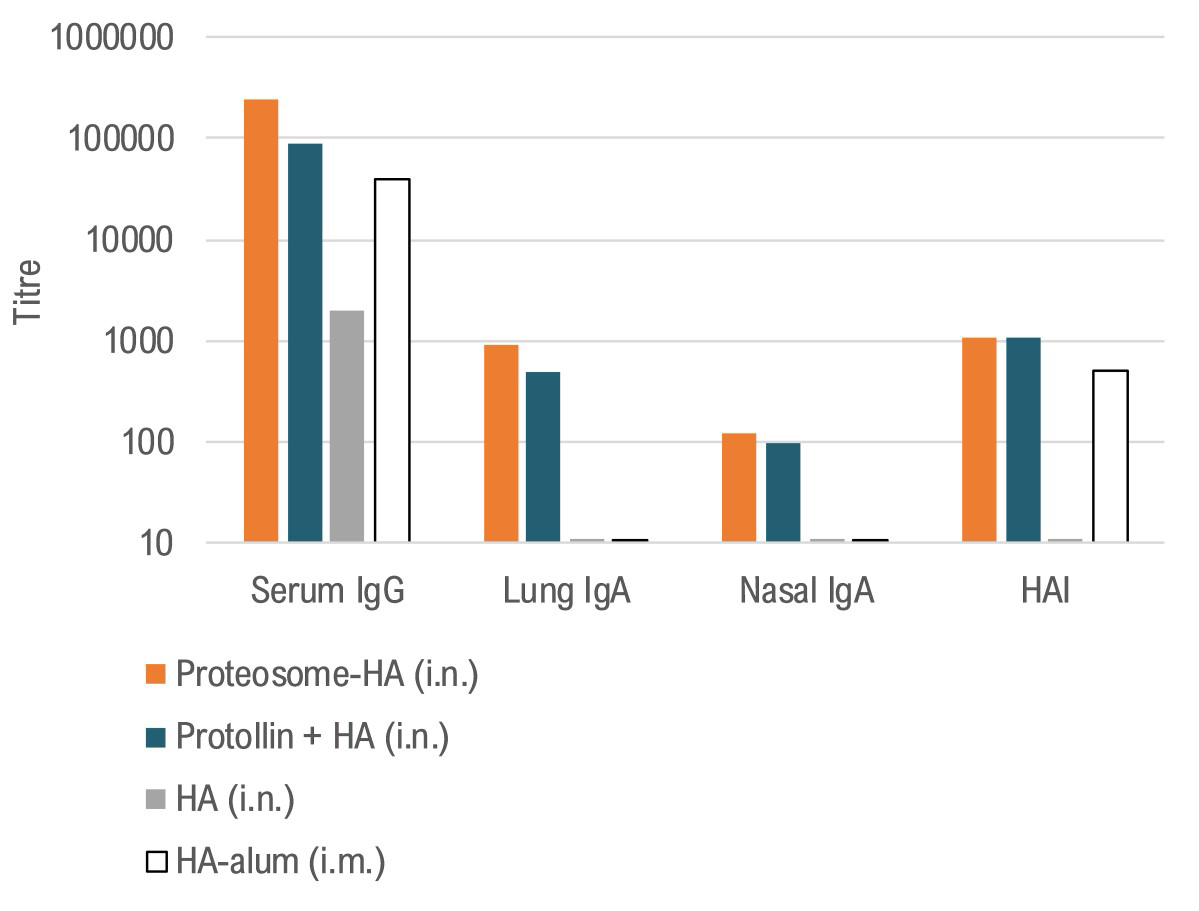
Nasal Influenza Vaccine - Preclinical immunology data with BDX200(Proteosome) and BDX100(Protollin)

Nasal Plague Vaccine - Preclinical immunology data with BDX100(Protollin)

Nasal Plague Vaccine - Preclinical efficacy data with BDX100(Protollin)